Publications
Selected publications.
2025
-
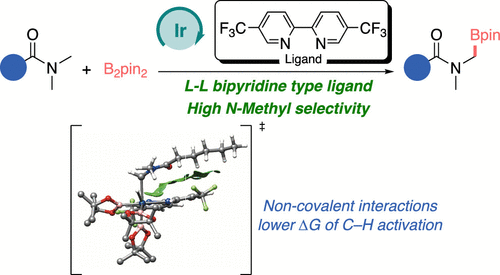 Bis-CF3-bipyridine Ligands for the Iridium-Catalyzed Borylation of N-MethylamidesDaniel Marcos-Atanes, Gonzalo Jiménez-Osés, and José L. MascareñasACS Catalysis, 2025
Bis-CF3-bipyridine Ligands for the Iridium-Catalyzed Borylation of N-MethylamidesDaniel Marcos-Atanes, Gonzalo Jiménez-Osés, and José L. MascareñasACS Catalysis, 2025Bipyridine and phenanthroline are well-established neutral ligands for promoting iridium-catalyzed borylations of aromatic C–H bonds. However, their use with aliphatic substrates is almost uncharted. Herein we demonstrate that introducing CF substituents at the 5- and 5′-positions of bipyridine generates ligands that enable an efficient and regioselective iridium-catalyzed borylation of the methyl group in a broad variety of methylamides. The reaction shows broad functional group tolerance and exhibits remarkable selectivity, offering a powerful approach for the borylation of challenging aliphatic C–H bonds. Mechanistic investigations, including computational analysis, suggest that the accelerating effect of the ligand is likely associated with the formation of non-covalent dispersion interactions between the carbonyl amide of the substrates and the trifluoromethylated pyridine rings of the ligand.
5-CF₃ bipyridine ligands boost Ir-catalyzed borylation of methylamides, enabling selective C–H activation at the methyl group through ligand–substrate dispersion interactions.
@article{doi:10.1021/acscatal.5c00933, author = {Marcos-Atanes, Daniel and Jim{\'e}nez-Os{\'e}s, Gonzalo and Mascare{\~n}as, Jos{\'e} L.}, title = {Bis-CF3-bipyridine Ligands for the Iridium-Catalyzed Borylation of N-Methylamides}, journal = {ACS Catalysis}, volume = {15}, number = {9}, pages = {7112-7120}, year = {2025}, doi = {10.1021/acscatal.5c00933}, url = {https://doi.org/10.1021/acscatal.5c00933}, eprint = {https://doi.org/10.1021/acscatal.5c00933}, }
2023
-
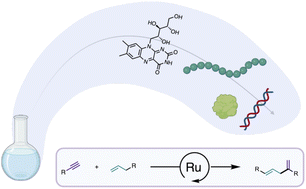 Ruthenium-catalyzed intermolecular alkene-alkyne couplings in biologically relevant mediaAlejandro Gutiérrez-González, Daniel Marcos-Atanes, Leonard G. Cool, and 2 more authorsChemical Science, 2023
Ruthenium-catalyzed intermolecular alkene-alkyne couplings in biologically relevant mediaAlejandro Gutiérrez-González, Daniel Marcos-Atanes, Leonard G. Cool, and 2 more authorsChemical Science, 2023Cationic cyclopentadienyl Ru(II) catalysts can efficiently promote mild intermolecular alkyne-alkene couplings in aqueous media, even in the presence of different biomolecular components, and in complex media like DMEM. The method can also be used for the derivatization of amino acids and peptides, therefore proposing a new way to label biomolecules with external tags. This C-C bond-forming reaction, based on simple alkene and alkyne reactants, can now be added to the toolbox of bioorthogonal reactions promoted by transition metal catalysts.
CpRu(II) complexes promote alkyne–alkene coupling in water and cell media, tolerating biomolecules and enabling peptide and amino acid labeling via bioorthogonal C–C bond formation.
@article{doi:10.1039/d3sc01254a, author = {Guti{\'e}rrez-Gonz{\'a}lez, Alejandro and Marcos-Atanes, Daniel and Cool, Leonard G. and L{\'o}pez, Fernando and Mascare{\~n}as, Jos{\'e} L.}, title = {Ruthenium-catalyzed intermolecular alkene-alkyne couplings in biologically relevant media}, year = {2023}, journal = {Chemical Science}, volume = {14}, number = {23}, pages = {6408 – 6413}, doi = {10.1039/d3sc01254a}, url = {https://doi.org/10.1039/D3SC01254A}, eprint = {https://doi.org/10.1039/D3SC01254A}, } -
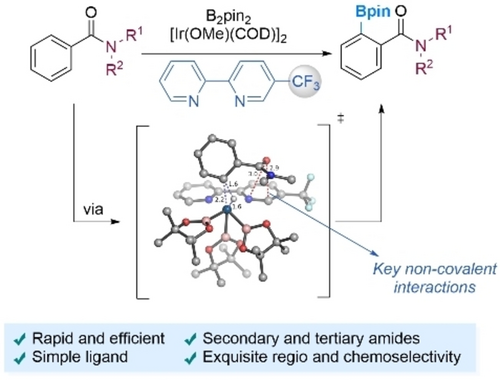 Iridium-Catalyzed ortho-Selective Borylation of Aromatic Amides Enabled by 5-Trifluoromethylated Bipyridine LigandsDaniel Marcos-Atanes, Cristian Vidal, Claudio D. Navo, and 3 more authorsAngewandte Chemie - International Edition, 2023
Iridium-Catalyzed ortho-Selective Borylation of Aromatic Amides Enabled by 5-Trifluoromethylated Bipyridine LigandsDaniel Marcos-Atanes, Cristian Vidal, Claudio D. Navo, and 3 more authorsAngewandte Chemie - International Edition, 2023Iridium-catalyzed borylations of aromatic C−H bonds are highly attractive transformations because of the diversification possibilities offered by the resulting boronates. These transformations are best carried out using bidentate bipyridine or phenanthroline ligands, and tend to be governed by steric factors, therefore resulting in the competitive functionalization of meta and/or para positions. We have now discovered that a subtle change in the bipyridine ligand, namely, the introduction of a CF3 substituent at position 5, enables a complete change of regioselectivity in the borylation of aromatic amides, allowing the synthesis of a wide variety of ortho-borylated derivatives. Importantly, thorough computational studies suggest that the exquisite regio- and chemoselectivity stems from unusual outer-sphere interactions between the amide group of the substrate and the CF3-substituted aryl ring of the bipyridine ligand.
A 5-CF₃ bipyridine ligand flips Ir-catalyzed borylation of aromatic amides from meta/para to ortho, driven by outer-sphere interactions between the ligand and the substrate’s amide.
@article{doi:10.1002/anie.202214510, author = {Marcos-Atanes, Daniel and Vidal, Cristian and Navo, Claudio D. and Peccati, Francesca and Jiménez-Osés, Gonzalo and Mascareñas, José L.}, title = {Iridium-Catalyzed ortho-Selective Borylation of Aromatic Amides Enabled by 5-Trifluoromethylated Bipyridine Ligands}, year = {2023}, journal = {Angewandte Chemie - International Edition}, volume = {62}, number = {18}, doi = {10.1002/anie.202214510}, url = {https://doi.org/10.1002/anie.202214510}, eprint = {https://doi.org/10.1002/anie.202214510}, }
2019
-
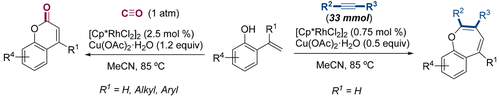 Practical, Large-Scale Preparation of Benzoxepines and Coumarins through Rhodium(III)-Catalyzed C-H Activation/Annulation ReactionsMoisés Gulías, Daniel Marcos-Atanes, José L. Mascareñas, and 1 more authorOrganic Process Research and Development, 2019
Practical, Large-Scale Preparation of Benzoxepines and Coumarins through Rhodium(III)-Catalyzed C-H Activation/Annulation ReactionsMoisés Gulías, Daniel Marcos-Atanes, José L. Mascareñas, and 1 more authorOrganic Process Research and Development, 2019Herein we disclose the assembly of benzoxepines and coumarins from 2-alkenylphenol precursors using [Cp*RhCl2]2 as the precatalyst and alkynes or carbon monoxide as reacting partners. The preparation of benzoxepines and coumarins can be scaled up to 33 mmol using low catalyst loadings.
Rh-catalyzed couplings convert 2-alkenylphenols into benzoxepines or coumarins using alkynes or CO, with scalable conditions (up to 33 mmol) and low catalyst loadings.
@article{doi:10.1021/acs.oprd.9b00191, author = {Gulías, Moisés and Marcos-Atanes, Daniel and Mascareñas, José L. and Font, Marc}, title = {Practical, Large-Scale Preparation of Benzoxepines and Coumarins through Rhodium(III)-Catalyzed C-H Activation/Annulation Reactions}, year = {2019}, journal = {Organic Process Research and Development}, volume = {23}, number = {8}, pages = {1669 – 1673}, doi = {10.1021/acs.oprd.9b00191}, url = {https://doi.org/10.1021/acs.oprd.9b00191}, eprint = {https://doi.org/10.1021/acs.oprd.9b00191}, }