Precision C–H Functionalization
Overriding steric bias in C–H functionalization using ligand-directed non-covalent interactions.
The Challenge: Overriding Sterics
In C–H activation, catalysts are lazy: they naturally react at the most accessible (least hindered) position. But mostly, the valuable chemistry happens at the crowded sites.
We changed the rules. Instead of fighting sterics with smaller ligands, we embraced non-covalent interactions. By designing ligands that "dock" onto the substrate via dispersion forces, we directed the Iridium catalyst exactly where we wanted it—even if it was the most crowded spot.
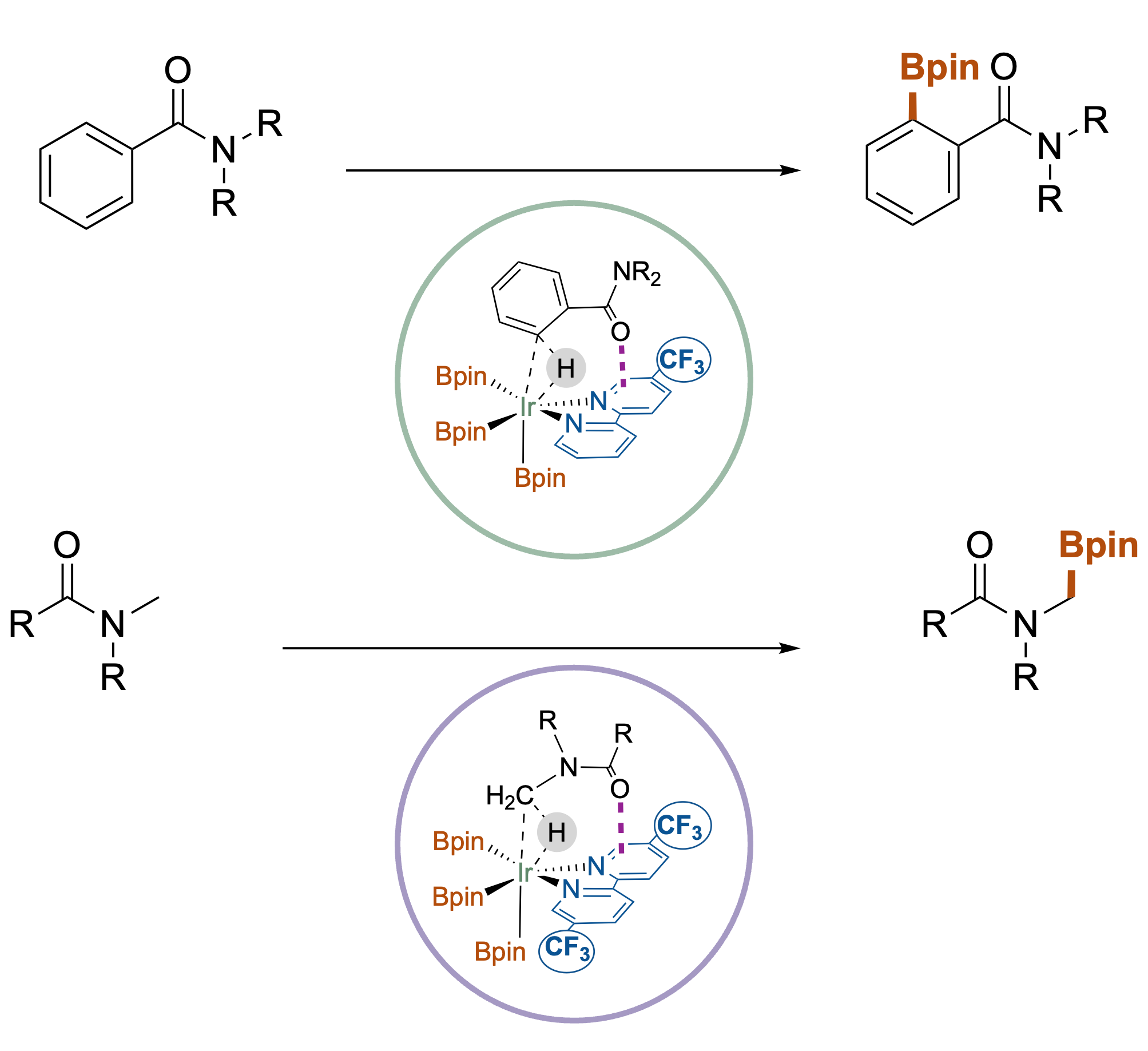
A Universe of New Molecules
This isn't just a niche reaction. By harnessing non-covalent interactions, we unlocked a vast chemical space, synthesizing a library of complex aromatic and aliphatic amides previously inaccessible.
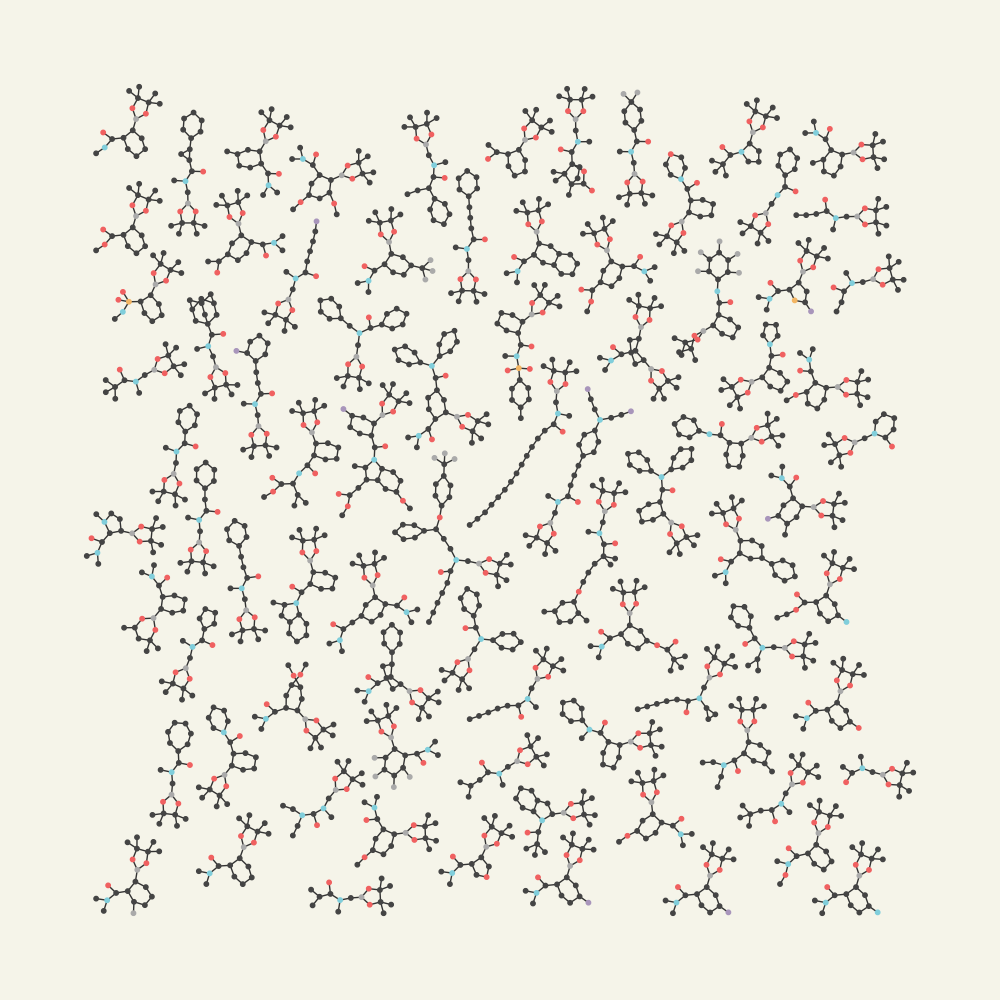
Dive Deeper into the Data
Static images only tell half the story. I have built an interactive web-viewer to explore the optimized geometries and transition states of the thesis library.
Launch 3D ViewerView structures directly in your browser. No software required.